
introduction on early-life stress
We all want to go back to times where we didn’t experience any stress - back to a careless childhood where the only problem ever was that summer recess was over too soon. But, what if you did experience severe stress as a child? Or even before that, if you grew up in a stressful environment as a baby or an infant? All of these examples can fall under the definition of early-life stress (ELS), where children experience physical, sexual, and emotional forms of abuse and/or neglect. More and more research shows that there is a link between ELS and psychological and physical problems later in life. According to the Center for Disease Control and Prevention, at least one in seven(!) children experience child abuse or neglect in the US, and this is probably an underestimate, since not all is reported. Moreover, research in animal models even shows evidence for being at a higher risk of developing Alzheimer’s disease or schizophrenia as a result of ELS. In this article, we’ll go deeper into the research, covering evidence from animal models, human data, and potential risks in neurodegenerative and psychiatric diseases.
Mechanisms at play
To start, we need to understand how stress influences your body and how it’s regulated in normal conditions. Here, research focuses on the hypothalamic-pituitary-adrenal (HPA) axis, the main mechanism involved in stress, among the sympathetic nervous system and inflammation-linked cytokines, but we’ll focus on the HPA axis for now. The HPA axis is a complex system between several hormone homes, such as the hypothalamus (brain), pituitary gland (brain), and adrenal gland (kidneys), hence the name. Its general function is to regulate stress reactions via many processes, where there are a few key hormonal players:

Very long story short, the HPA axis regulates and reacts to stress by releasing these hormones, which cause several other bodily systems to react to the stressor. For example, they influence the dilation or constriction of blood vessels*, impact your sleep/awake cycle, release glucose as part of your metabolic system, and suppress the immune system. All these processes make sure you can react properly to stress by ‘fight or flight’-ing.
Experiencing prolonged and more extreme levels of stress can make your HPA axis more hyperactive and sensitive. You can probably imagine that this could also make one more vulnerable to stress, although this also depends on the type and the severity of the stressor(1). Cortisol impacts many bodily systems, such as glucose availability as part of your energy metabolism and the immune system, but can also have damaging effects on the hippocampus, our memory brain region. Even if we experienced similar situations in the past, high stress levels in present situations make us ‘forget’ how we can overcome these difficult situations. We therefore handle stress not as well, while also increasing chances of anxiety and other stress-related disorders (2).
However, it’s crucial to note that not all stress is bad; your body is incredibly good at handling mild to moderate stressors in life. Studies also show that a bit of stress throughout life helps to regulate the HPA system better, making you more resilient to stress. Stress is part of our life, and our minds and bodies are built to deal with stress; that’s why we have this amazing and well-functioning HPA-axis.
What science shows us
When our well-functioning HPA-axis stops being so well-functioning, things start to get less great. A lot of research evidence shows that the dysregulation of the HPA axis increases the risk of depression, schizophrenia, and memory dysfunction. In a maternal deprivation model of ELS in mice, where pups are separated from their mother over a few weeks at random intervals, research shows a longer lasting increase in cortisol in the pups (3). These higher levels of cortisol during development can impair the regulation of cortisol later in life, and make them more sensitive to stress in adulthood.
Maternal deprivation is one of several ways to mimic ELS in an animal model. Other models are (mal)nutrition, limited bedding, grooming, or nesting, and maternal stress. One famous, but nowadays ethically questionable, experiment from Harry Harlow in the 1960’s showcased the power of maternal attachment (4). In this experiment, Harlow separated a rhesus monkey mother and infant from each other, and posed two mom-shaped mannequins to the infant (image 2): one “mom” (left) provided only the physiological needs, such as food and water. The other “mommy” (right) provided something else: warmth and attachment, without food and water. This experiment showed that the monkey infant strongly prefers the mommy that provides warmth and attachment. The monkey would only get water and food if necessary from mom on the left, and would then return back to the other mom. This shows how powerful maternal attachment can be, and we even saw this in a slightly different setting recently over social media, where Punch the infant monkey found a plushie ‘surrogate’ mom, to which he constantly clings to.

In the many animal studies that have been conducted on ELS, there is evidence to support that ELS has an effect later in life on the gut microbiome (5), insulin metabolism, hypertension (6), food choices (7), emotional learning (8), long-term cortisol regulation and even sexual maturation & behavior (9-10). Research even finds some sex differences as a result of ELS - it increases fat intake in females, since ELS alters specific brain circuits that are responsible for food intake and food choices. In an animal model of Alzheimer’s disease (AD), researchers found that negative early life experiences increased the risk of developing AD, while positive early life experiences were related to resilience against developing AD. They found that ELS changes the programming of the HPA axis, influences inflammation in the brain, negatively impacts brain plasticity, and affects cellular function.
Enlightening Epigenetics
When we look at maternal stress as a model of ELS, things become especially interesting. You may think, how can a mother’s stress level during pregnancy impact how the offspring copes with stress? This is where the topic of epigenetics comes into play, i.e. how traits can travel over generations, embedded in and around genes. In short, epigenetics influences gene expression without actually changing the genes, but rather making a part of a gene more or less accessible to be expressed by changing its 3D structure. Imagine this: your genetic code is like a cookbook you got from your parents. They have their own cookbook, including sticky notes, scribbles in the margin, bookmarks, tears and wears - these are the epigenetic regulators - and copied a book for you with their stuff combined. Some epigenetics, like the sticky notes, don’t get copied (or inherited), while some, like scribbles, do get copied to your cookbook. If we then circle back to stress or malnutrition in the mother, can you imagine how these scribbles and tears in her cookbook may be copied into yours?
A review from 2015 covered many of the epigenetic aspects of ELS (11). Anxiety or depression experienced during the 3rd trimester in pregnancy were correlated with higher levels of stress response in infants at 3 months of age. Moreover, maternal depression during the 2nd trimester was associated with epigenetic changes in the offspring in serotonin genes and in white blood cells. Even low socioeconomic status can be used as an indicator of ELS, as this can result in poor quality of dietary intake and higher physical workload. Here, we even see epigenetic changes in blood associated with low socioeconomic status. To make things even more interesting, there are consistent findings on the delivery method during labor, such as the cesarean section (C-section) or a vaginal birth, and epigenetic effects in infants. Infants delivered by a C-section experience more stress than infants born by vaginal delivery; the latter seem to adapt better to new conditions, and seem less prone to metabolic syndrome and asthma later in life. Here, we do see epigenetic differences in the immune response, especially in white blood cells.
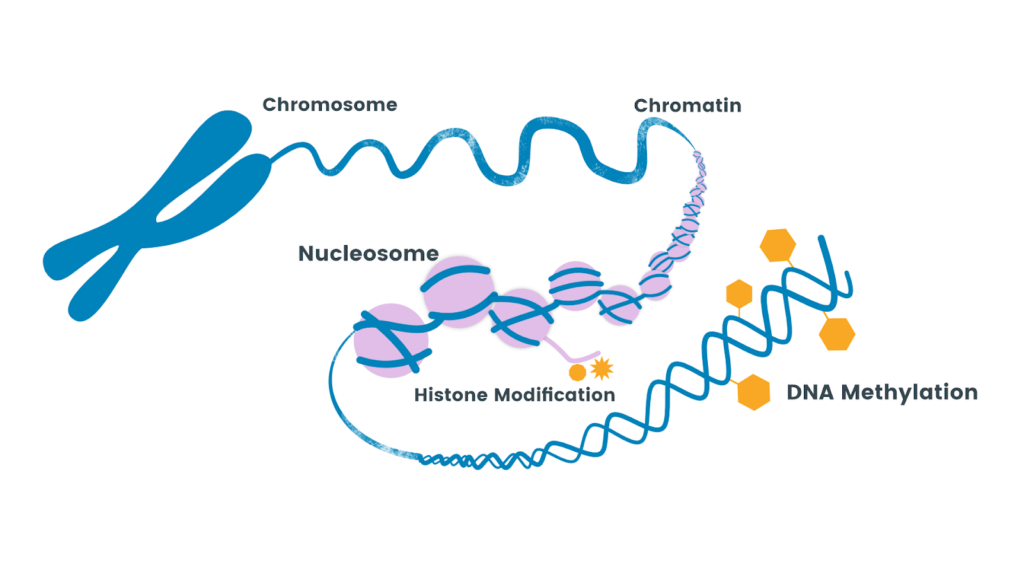
In animal models, the meta-analysis from Fogelman & Canli (2019) showcases a beautiful overview of research on ELS and (epi)genetics in animal models (12). They state that the majority of research suggests the role of DNA methylation as an epigenetic mechanism. In short, DNA methylation happens when a methyl (CH3) group binds to specific amino acid sites on the DNA. When a methyl group binds to the DNA, the structure of the chromatin changes, making the DNA more or less accessible to be translated or ‘expressed’ into an active protein (13). ELS has been linked to methylation, meaning ELS can impact gene expression, and several studies confirm this. To highlight a study from this review, a study published in Nature from Weaver et al. (2004), showed that poor care from a rat mother to her pups led to higher methylation in stress-related receptors in the pups’ hippocampus, our memory center. The methylation decreased expression of this gene, making the pups less able to regulate stress and therefore making them more reactive to stress, even into adulthood.
Will I be lost forever?
Although ELS can sound scary and inescapable, it is also important to keep in mind that not every traumatic event may harm you for life. Your brain is dynamic, especially throughout childhood and adolescence, and can still adapt throughout adulthood. Not all forms of childhood trauma have the same negative effects, and especially nowadays, there is a tendency to label any unpleasant event as traumatic, from divorced parents to moving houses. That is not to say that these events cannot be traumatic, but it’s important to really define what early-life stress and actual childhood trauma is. Dutch psychiatrist and professor Christiaan Vinkers highlights this in his book ‘Littekens uit je jeugd’ (“Scars from your childhood, red.), where Vinkers dives deep into what childhood trauma is and what it’s not, the mechanisms at play, and how to work towards healing and recovery.
To wrap up on a brighter note: it’s never too late to make friends with your own stress. Our early baggage might shape us, but it doesn’t have to steer the whole journey. Learning to spot rising tension, knowing how it messes with your body and mood, and experimenting with ways to reset; that’s the real trick to growing tougher skins and calmer minds. Because the goal isn’t to banish stress (good luck with that), but to get better at dancing with it. And if you ever wonder where your stress habits started, well, nature and nurture probably deserve joint custody.
Author: Lotte Smit
References